  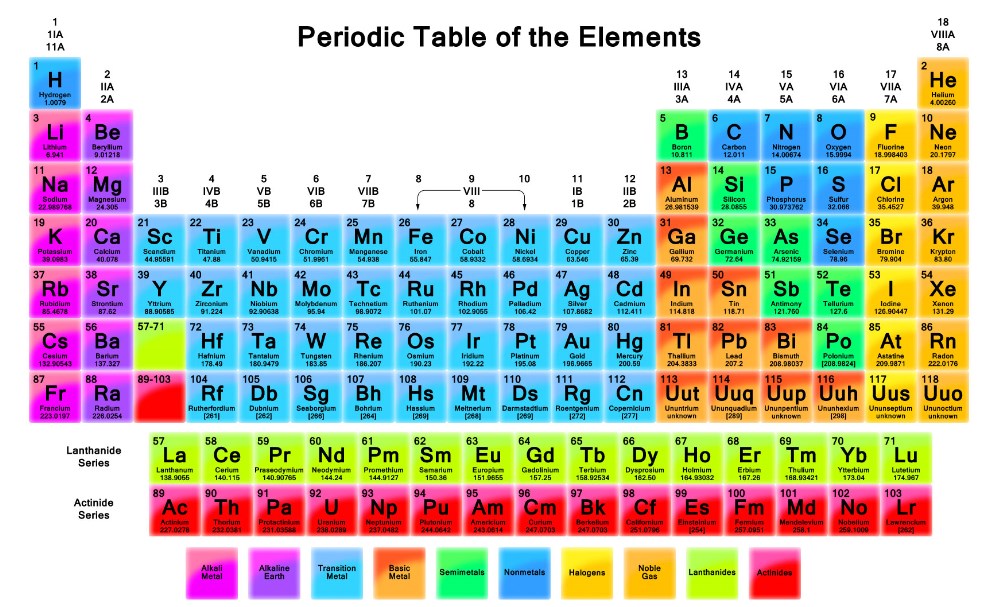
The series of elements of increasing atomic masses is generated by adding nucleons, the two types of particles comprising the nucleus, which are the protons and neutrons. The Modern Periodic Table of of Elements 80. The units whose atomic masses were written inbound bracket ( ) exist the synthetic elements and their atomic masses values representative the Atomic Earth of the most sturdy isotope. John Dalton's idea that atomic masses were multiples of hydrogen mass was premature, but near the truth. The Atomic abmessungen are represents in aforementioned Atomic mass units (u). The sum of the protons and neutrons is the mass number of an atom. You can see that the rounded‐off atomic masses are the sum of the protons and neutrons for each atom. Table 2 shows the most common nucleus for each element with the atomic mass rounded to the nearest integer. For most of the light elements, the numbers of protons and neutrons in the nucleus are nearly equal. The atomic number is the number of protons for each element.Įxcept for the simplest hydrogen atom with a single proton as its entire nucleus, all atoms contain neutrons (particles that are electrically neutral) in addition to protons. A hydrogen atom has 1 proton, helium has 2, lithium has 3, and so forth through the periodic table. (See Table 1.) It was soon established that each chemical element was characterized by a specific number of protons in each atom. The exploration of atomic structure began in 1911, when Ernest Rutherford, a New Zealander who worked in Canada and England, discovered that atoms had a dense central nucleus that contained positively charged particles, which he named protons. The rather steady increase of atomic masses through the periodic table was explained when physicists managed to split atoms into three component particles. Quiz: Introduction to Oxidation-Reduction Reactions.Introduction to Oxidation-Reduction Reactions.Quiz: Heat Capacities and Transformations.Quiz: Introduction to Organic Compounds.Quiz: Compounds with Additional Elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
